Researchers at Technion have developed a first-of-its-kind 3D implant that combines muscle and fat tissue with both blood vessels and a lymphatic network, a step they say could eventually help treat patients with major tissue loss from injuries, burns or cancer surgery.
The findings were published in the journal Cell Biomaterials by an international team led by the Levenberg Laboratory in the Faculty of Biomedical Engineering at the Technion-Israel Institute of Technology.
1 View gallery
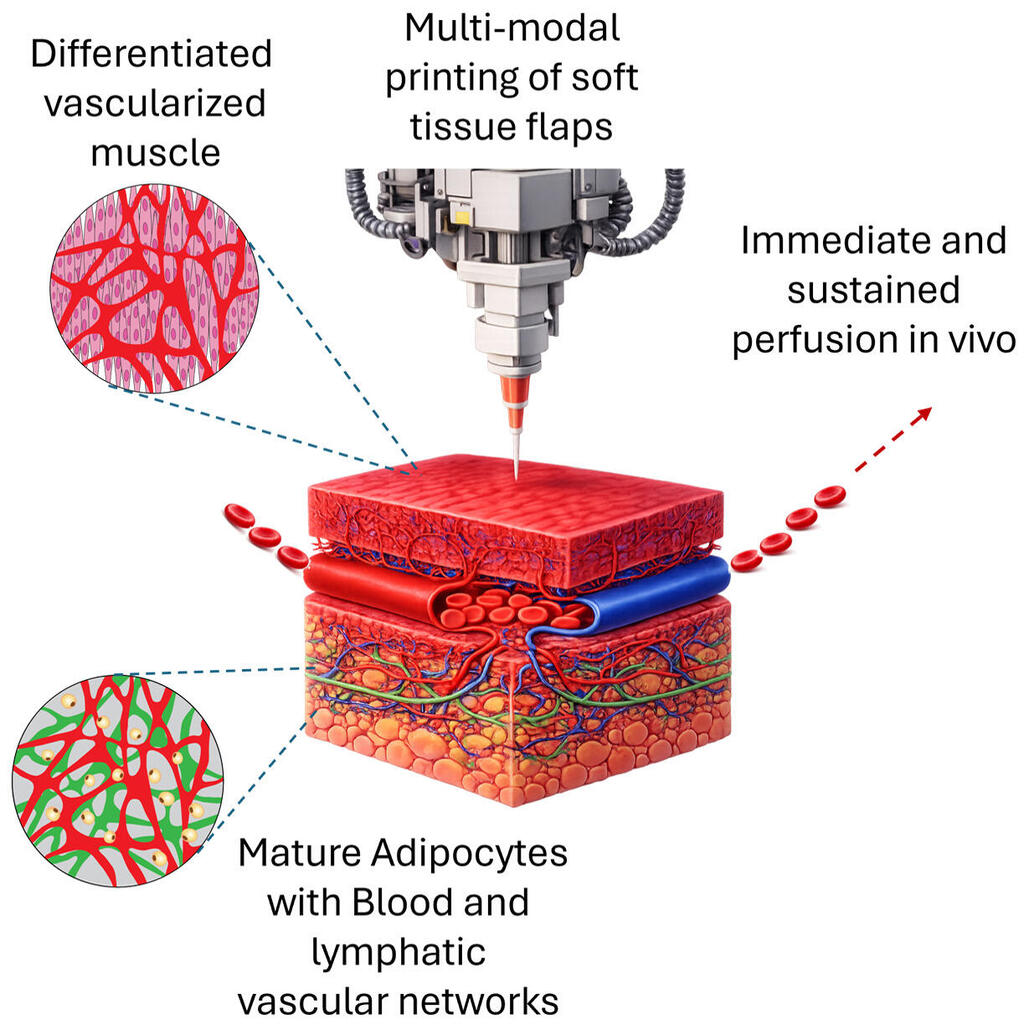

Graphic abstract (from bottom left, clockwise): Muscle tissue containing mature cells and vascular networks; fat tissue containing blood vessels and lymphatic networks; printing of these flaps; and immediate perfusion of the flap by the host tissue
(Illustration: Technion)
The current standard treatment for significant tissue loss is an autologous flap, in which tissue is taken from one part of a patient’s body and transplanted to the injured area. Doctors use that method because tissue taken from another person can trigger immune rejection and other complications.
Prof. Shulamit Levenberg, who heads the Technion research group, said the new development could offer a different path.
“Our development represents a significant step toward the production of complex implantable tissues for cases involving loss of muscle and fat tissue due to injuries, burns, tumor resection, and more,” she said. “The technology presented in the paper may, in the future, enable the production of personalized flaps tailored to the specific characteristics of an individual patient’s injury.”
 Prof. Shulamit Levenberg Photo: Technion
Prof. Shulamit Levenberg Photo: TechnionA flap is a tissue implant that includes a vascular system, which helps it connect to the damaged site after implantation. Without such a system, the implant does not immediately receive enough oxygen and nutrients, and waste is not efficiently cleared away.
According to the researchers, the new flap is the first to incorporate not only a hierarchical blood vessel network but also a lymphatic network, which is essential for draining fluids from the spaces between cells. The flap also includes muscle and fat tissues in a single structure, along with an arteriovenous loop designed to connect directly to the blood supply at the implantation site.
The team said the overall construct closely mimics natural tissue, including the extracellular matrix, or ECM, the support structure surrounding cells. They said that required optimizing the printing process, both in the design of the printed structure and in the calibration of the syringes used to place cells within the flap.
 Dr. Eliana Fischer Photo: Technion
Dr. Eliana Fischer Photo: TechnionIn experiments in rats, the researchers said connecting the flap to the target site led to rapid integration. They reported continuous delivery of oxygen and nutrients, stable blood flow, normal muscle development and stability of the fat cells. The result, they said, was that the flap became an integral part of the implantation site and surrounding tissue, both aesthetically and functionally.
The study was led by Levenberg, who heads the Technion’s Laboratory for Tissue Engineering and Stem Cells, along with Dr. Eliana Fischer, a physician and graduate of the Technion’s Ruth and Bruce Rappaport Faculty of Medicine who is now a Ph.D. student in the lab, and doctoral student Anna Tsukerman, a biology graduate who entered the program after six years in the biomedical industry.
The researchers used a bio-ink based on components of the extracellular matrix, which they said mimics the natural tissue environment and makes it possible to print muscle and fat tissue in a way that encourages cell differentiation.
 Anna Tsukerman Photo: Technion
Anna Tsukerman Photo: TechnionTo support growth of the vascular system, the team printed engineered blood vessels and developed a bioreactor in which they were cultured under flow conditions meant to mimic natural blood flow and promote maturation of the endothelial layer.
In the next stage, the flaps were implanted in rats and directly connected to an artery and vein at the target site. Although the experiments were conducted in rats, the engineered tissues were made from human cells in order to test the technology’s feasibility for eventual use in people, the researchers said.
The group has now begun testing the technology in large animals, which it described as the next step toward possible clinical trials.
The study also included researchers from the Department of Plastic Surgery at the School of Medical Sciences at Tokushima University in Japan. It was supported by the European Union through an ERC grant and by the Japanese company Nichia under a joint grant program involving Nichia, the Technion and Tokushima University.

