Professor Shulamit Michaeli is the recipient of the 2026 Israel Prize in Life Sciences Research. She will receive the award on the 78th Independence Day, in recognition of her contributions to the study of the RNA of infectious disease pathogens. “It’s very exciting,” Michaeli said in an interview with the Davidson Institute.
A professor of microbiology at Bar-Ilan University, Michaeli is a pioneer in parasite Ribonucleic acid (RNA) research. Her work focuses on parasites that cause serious diseases, including cutaneous leishmaniasis, which is caused by the leishmania parasite. For many years, she has studied how these parasites infect their hosts and communicate, in the hope of developing an effective treatment.
The Girl Who Dreamed of Becoming Marie Curie
Michaeli, 70, grew up in Givatayim and was honored in 2022 as one of the 100 most influential women to come out of the city, as part of its centennial celebrations. “I come from a scholarly family, originally a family of rabbis and judges, but when it came to biology—life sciences—I had no one to guide me in that direction. I was drawn to it on my own; it fascinated me,” she says. “In high school I studied biology, and it felt only natural to continue on to life sciences.”
Her father was one of the founders of the Israeli Combat Engineering Corps and established the IDF’s heavy mechanical equipment unit. “I was drawn to science from a very, very young age,” Michaeli told the Davidson Institute. “I dreamed of being Marie Curie, of having a laboratory next to my house, and of getting up in the middle of the night to check on my experiments.”
Michaeli completed her bachelor’s degree with honors at Tel Aviv University. “I was always drawn to the life sciences, especially where they intersect with chemistry,” she said. “I was among the first students to go straight into a direct doctoral track at Tel Aviv University.” During her doctoral studies, she researched gene activity in bacteria, but by the end of her PhD she wanted to work on organisms more complex than bacteria.
“At exactly that time, molecular biology was beginning to take shape, along with the study of diseases that science and medicine had largely neglected until then, even though they affect millions of people.” The organism she chose was a single-celled parasite. “It was really an ideal system for me,” she said, “because it introduced me both to the world of parasites and to a major infectious disease.”
Michaeli went on to do her postdoctoral work at the University of San Francisco, where she was introduced to the field of RNA research—molecules involved in many processes inside the cell, especially in carrying out the instructions encoded in DNA and translating them into the proteins the cell produces. During this time, she participated in the discovery of a process known as trans-splicing, in which a parasite’s RNA molecules are cut and rejoined to enable the production of specific proteins.
In 1990, Michaeli established her own laboratory at the Weizmann Institute of Science. At the time, she says, parasitic RNA “was not considered a particularly hot topic, and people did not really understand its importance. They thought the most important things were DNA, because it is the hereditary material itself, and proteins, because they do the actual work. RNA was seen merely as an intermediary. But I understood that it was important, and I stayed with it throughout my professional life.”
A single small change can alter the entire structure of a parasite’s ribosome. The leishmania parasite undergoes changes during its life cycle as it moves from the sand fly to a mammalian host.
3 View gallery
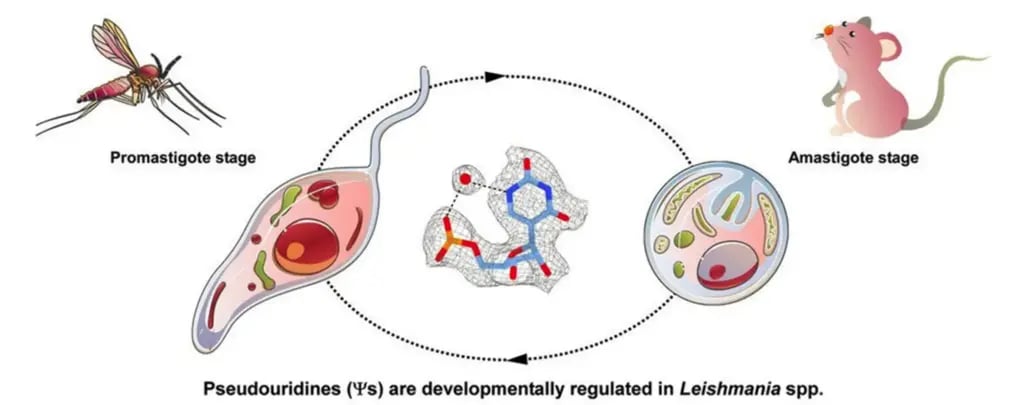

A single small change can alter the entire structure of a parasite’s ribosome. The leishmania parasite undergoes changes during its life cycle as it moves from the sand fly to a mammalian host
(Rajan, K. S. et al. Cell Reports (2024))
Starting Over
In 1998, Michaeli moved to Bar-Ilan University. “I started my life over, and fortunately I was very successful,” she said. Even after the move, she remained closely connected to her colleagues at the Weizmann Institute and continues to collaborate with researchers there. In a study she recently published with Nobel Prize laureate Ada Yonath of the Weizmann Institute of Science, who deciphered the structure of the ribosome—the cell’s protein-making machinery—the two found that the ribosome of the leishmania parasite adapts to changes in the parasite’s environment.
The parasite moves from the gut of the female sand fly to the bloodstream of the mammal she bites. That transition involves a major shift in temperature, from about 27 degrees Celsius in the fly’s gut to roughly 37 degrees in the mammalian body. To survive in its new environment, the parasite must adapt, and it does so through unique chemical changes in its ribosomes. Michaeli and Yonath showed that even a single such change can alter the ribosome’s entire structure, and with it the range of proteins it can produce. “We were among the first to show that these adaptations change as the parasite moves between its two life stages, and that this is what allows its ribosomes to function at such different temperatures,” Michaeli said.
Michaeli also discovered a unique mechanism by which RNA molecules inside the nuclei of trypanosoma parasites, which cause sleeping sickness, and leishmania parasites are silenced. Known as RNA interference, or RNAi, the mechanism involves a specific class of small RNA molecules. These molecules bind to messenger RNA, which carries DNA’s protein-coding instructions to the ribosome, and prevent the production of certain proteins when they are not needed. “During my career, I was among the first to argue that RNAi exists not only in the cell’s cytoplasm but also in the nucleus itself, and we called it snoRNAi”—a form of small RNA interference found in the nucleolus, a structure within the cell nucleus.
More recently, Michaeli discovered a distinctive stress-response mechanism based on this process: when conditions become unfavorable, the parasite can trigger its own death. It does so by shutting down the production of messenger RNA molecules, which carry the instructions needed for producing the proteins essential for its survival and function. To achieve this, the parasite halts the RNA splicing machinery, effectively setting off a self-destruct process. Michaeli also found that certain drugs can trigger this self-destruction mechanism, and she is now working to understand how the parasite carries out this self-destruction and which molecules are involved.
The molecule Michaeli and her colleagues discovered sends the parasite into a dormant state.
3 View gallery

The molecule Michaeli and her colleagues discovered sends the parasite into a dormant state. Illustration of a trypanosoma parasite in the bloodstream
(Illustration: Kateryna Kon/ Shutterstock)
Speaking the Parasites’ Language
Michaeli recently discovered a type of long RNA molecule, known as lncRNA, that determines when a trypanosoma parasite changes into the form that allows it to infect humans. The molecule also enables the parasites to move together as a group. This ability is essential, since the parasites must travel from the gut of the tsetse fly (Glossina) to its salivary glands, from which they are transmitted to humans through a bite. The study showed that when this molecule is missing, the parasites can no longer move together, preventing them from spreading the disease. It was the first study to define the role of this type of RNA in the parasite’s reproductive cycle.
She also found that the parasites pass information to one another through exosomes—tiny vesicles containing RNA and proteins that they secrete to warn each other of stress. “I am trying to understand their language, and how they pass messages to other parasites,” Michaeli said. “We showed that if they are in poor condition, they secrete exosomes that tell the other parasites, ‘Don’t take us on the long journey; we’re not okay.’ I am still trying to understand what in the exosomes conveys that message.”
In another study, Michaeli and her colleagues identified an RNA molecule unique to the parasite. “We have an amazing study, in collaboration with a group from Portugal, showing that a single small nucleolar RNA molecule can cause the sleeping sickness parasite to stop infecting,” Michaeli said. The molecule they discovered, also a type of lncRNA, contains a small RNA molecule whose role is to shut down the activity of messenger RNA molecules involved in the parasite’s development.
When the parasite infects a person, it multiplies rapidly in the bloodstream and causes severe disease. At a certain stage, however, it stops reproducing in order to complete its life cycle, remaining in the patient’s blood until another fly bites the person and ingests the parasite-rich blood. It turns out that this newly discovered molecule is responsible for shifting the parasite into a dormant state. Increasing production of the molecule can therefore force the parasites into dormancy. In this state, they are unable to reproduce, and the active infection in the mammalian host is halted. “We are now continuing this research, and soon we will write a groundbreaking paper on the subject,” she concludes.
Not Slowing Down
Michaeli says her lab is currently developing a treatment for cutaneous leishmaniasis based on a nanoparticle that penetrates the parasite’s cell and destroys it within twenty minutes. The treatment has already shown promise in animal studies, and her team is now working with Tel Aviv Sourasky Medical Center to adapt it for human use. The next step will be clinical trials to test whether it is safe and effective in people. If successful, it could make a meaningful difference for communities affected by cutaneous leishmaniasis across parts of the Middle East, Africa, Asia and Latin America.
“Today, looking back, I can say that my long-term contribution to deciphering the highly unique pathways of RNA biology in trypanosoma parasites is being recognized,” Michaeli says. “For example, the understanding that RNA can be modified after it is produced emerged from studies on trypanosomes.”
“People ask me, ‘Shula, you’ve already received the Israel Prize—maybe it’s time to slow down a little?’ But I never worked for a prize. I worked because I love what I do. Even when I teach students and young researchers, it is important to me to pass on that love of microbiology and RNA. I’ve helped shape an entire generation of RNA researchers in Israel.”
Winning the prize has not slowed her down. If anything, her passion for the work continues to grow. “I have research grants and a lab full of students, and I feel as though I’m only just getting started. My love for this field only deepens with time, because today’s technology makes it possible to do such extraordinary things that it’s impossible to stop. This is an exceptionally exciting time for science.”


