The Health Ministry's expert panel on Wednesday voted with an overwhelming majority in favor of administering Pfizer's and BioNTech's pediatric COVID-19 vaccines to children aged 5-11 and the inoculation drive for that age group is set to take off over the next week or two, but some doubts remain hanging in the air:
The ministry's pandemic task force voted almost unanimously in favor of recommending vaccinating children aged 5–11, but panel members were divided on whether to also inoculate children who have already recovered from the disease.
3 View gallery


Members of the Health Ministry's expert panel on COVID-19 vaccine for children giving a presser, November 10, 2021
(Photo: Yair Sagi)
While 23 experts voted in favor, 34 others voted in favor but with reservations about the date of recovery and eight others objected outright.
In addition, it remains unclear how far apart will both vaccine doses be administered. Pfizer recommends administering both doses of its vaccine three weeks apart, but the Health Ministry said that it will further discuss the issue as data is still insufficient at this point.
Some have decried the board for lack of transparency, with critics claiming the vote was held behind closed doors to keep the public from hearing opinions of experts that may discourage vaccination.
Hillel Yaffe Medical Center's Director of the Infectious Diseases Unit Dr. Michal Stein told the Ynet studio that none of the attendants at the discussion objected to vaccinating children outright and deliberations touched on whether to roll out the vaccine immediately or wait for data from the United States, which rolled out COVID-19 vaccines for children late last week.
"A professional discussion should be closed so that people can express their opinions freely. The discussion was very to the point. People have expressed their opinion on questions like whether to recommend the vaccine or ‘allow’ it, which is more nuanced," Stein said.
"We discussed a vaccine for those who have recovered and opinions on the issue were pretty evenly split, although in the United States the recommendation was to vaccinate recovering patients. In the meantime, we are waiting and we will discuss the again [on Friday]."
Dr. Stein added that the expert panel had taken into account the possibility of some children experiencing adverse reactions due to the vaccine, but no such cases have been yet reported."
"Nothing severe has been reported. Obviously, there will be side effects, this is a vaccine, not a lollipop. The question is whether the benefit significantly outweighs the risk," she said. "A wise doctor once taught me that what has no side effects, has no effects at all."
3 View gallery
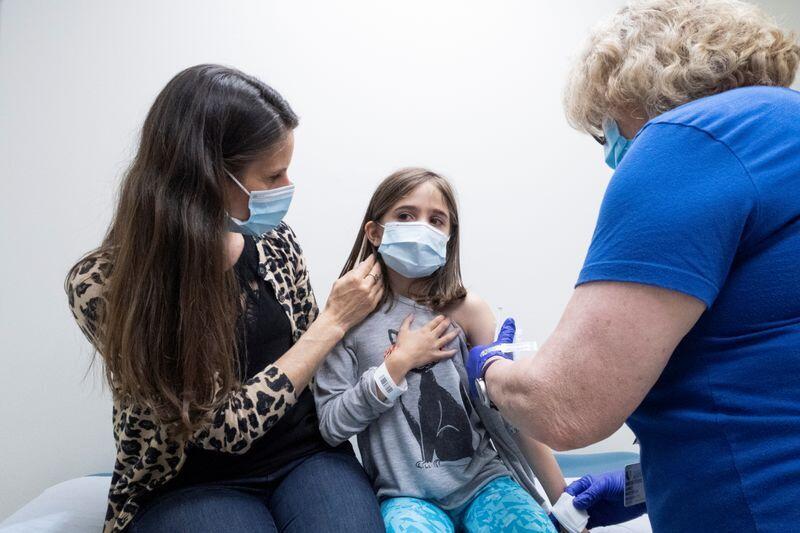

A young girl receives the coronavirus vaccine during clinical trials to test its safety and efficacy in children
(Photo: Reuters)
After the panel's decision, it is now up to Health Ministry Director-General to sign off on an order to roll out the COVID-19 vaccine for children.
Children are expected to receive only a third of the amount adults received with every vaccine dose three weeks apart.