Mira Bar-Matthews, a researcher at the Geological Survey of Israel (GSI), was presented with the 2026 Israel Prize in Geology and Earth Sciences for her exceptional contribution to the study of past climates.
Throughout her career, Bar-Matthews has focused on cave deposits or speleothems, including stalactites and stalagmites, which she and her team have used as “natural archives” to reconstruct climate conditions and earthquake activity over tens to hundreds of thousands of years.
5 View gallery


Bar-Matthews was among the first to demonstrate how detailed information about past climate conditions and precipitation patterns can be extracted from natural archives. Bar-Matthews with a stalactite
(Photo: Anton Vox)
At a time when climate change is unfolding before our eyes, the study of Earth’s ancient climate is not merely an academic pursuit, but a vital tool for understanding the planet’s future. Bar-Matthews was among the first to show how nature can preserve detailed records of past climate conditions and precipitation patterns.
She later demonstrated that these same materials can also reveal evidence of ancient earthquakes and ground movements in the distant past. By combining advanced geochemical methods, she opened a window onto the environmental and tectonic history of the Mediterranean and the Middle East.
Stories in stone
Bar-Matthews was born and raised in Jerusalem’s Beit Hakerem neighborhood. She began her academic journey in 1969, studying geology and developing an interest in the processes that shape the Earth over time. She continued her studies in the Department of Geology at the Hebrew University of Jerusalem and later joined the Geological Survey of Israel as a researcher.
Over the course of her scientific career, Bar-Matthews investigated how water reacts with rocks and alters their composition, particularly in the formation of carbonate deposits. Carbonate is a compound formed through a reaction between carbon dioxide dissolved in water and substances in the surrounding environment. One of the most common carbonate compounds in nature is calcium carbonate, the main component of limestone and many other carbonate deposits.
5 View gallery


Rainwater absorbs carbon dioxide from the atmosphere and the soil, becomes slightly acidic, and dissolves calcium from rocks. When environmental conditions change, the calcium carbonate precipitates and recrystallizes. Illustration of the water cycle and the formation of stalactites in caves
(Photo: Gary Hincks / Science Photo Library)
Carbonate rocks form in a range of environments and through different processes. In the sea, many are formed through biological activity: marine organisms build shells and skeletons from calcium carbonate, and after they die, their remains accumulate to form layers of rock. On land, by contrast, the process is mainly chemical. Rainwater absorbs carbon dioxide from the atmosphere and the soil, becomes slightly acidic and dissolves calcium from rocks.
When environmental conditions change, for example because of shifts in temperature or evaporation, the calcium carbonate precipitates and recrystallizes. In this way, a variety of terrestrial carbonate deposits are formed, including stalactites and stalagmites in caves, travertine in springs and more.
One of the main uses of carbonate deposits in research lies in their ability to preserve a “memory” of the environmental conditions at the time they formed. The chemical composition of the carbonate changes according to the conditions under which the mineral precipitated. A great deal of information can be derived from the ratio between different isotopes: isotopes are different forms of the same element that differ in the number of neutrons in the nucleus. Different isotopes behave the same way in chemical reactions, but they can be distinguished by their weight. In carbonate deposits one can find oxygen isotopes—oxygen-18 alongside the more common oxygen-16—and carbon isotopes—carbon-13 together with the more common carbon-12.
One of the main scientific uses of carbonate deposits lies in their ability to preserve a geochemical “memory” of the environmental conditions in which they formed. In particular, the isotopic composition of carbonate can reflect the conditions under which the mineral precipitated. A great deal of information can be derived from the ratios of different isotopes. Isotopes are forms of the same element that differ in the number of neutrons in the nucleus. They have nearly identical chemical behavior, but can be distinguished by their mass. In carbonate deposits, researchers commonly analyze oxygen isotopes – oxygen-18 alongside the more abundant oxygen-16 – and carbon isotopes – carbon-13 together with the more abundant carbon-12.
5 View gallery
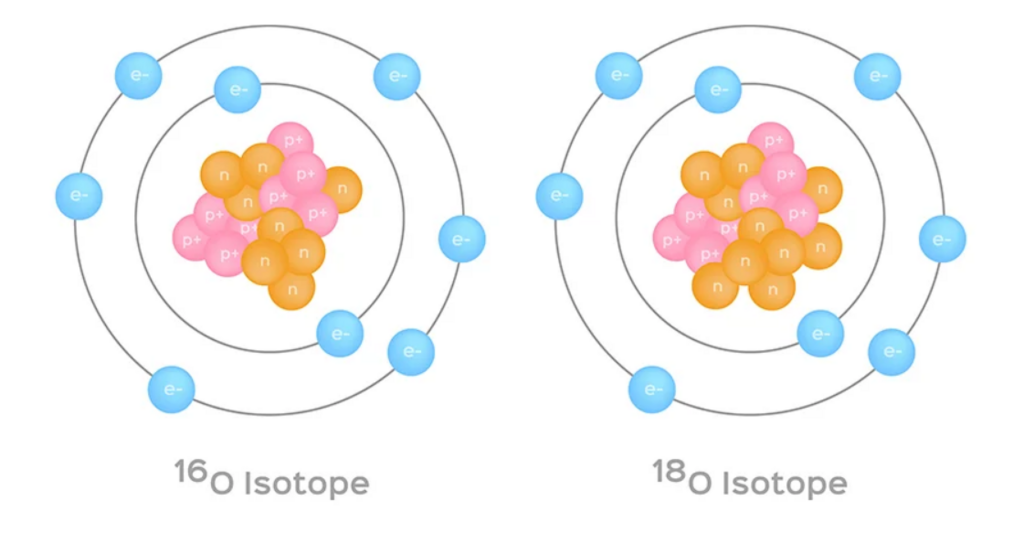

Isotopes are different forms of the same element, differing in the number of neutrons in the nucleus. Illustration of two oxygen isotopes, oxygen-16 and oxygen-18
(Photo: Gary Hincks / Science Photo LibraryShutterstock, Gritsalak Karalak)
The ratio between oxygen isotopes, for example, can reflect the isotopic composition of the rainwater from which a carbonate deposit formed and may preserve information about changes in precipitation, evaporation, temperature and broader hydrological conditions over time. The same principle underlies studies of polar ice layers, which have been used to reconstruct glacial and interglacial periods over hundreds of thousands of years. Just as ice preserves information about past climate through oxygen isotope ratios, carbonate deposits likewise preserve evidence of climatic and environmental conditions in the past.
When Bar-Matthews began her research, the study of past climate relied mainly on polar ice deposits and marine sediments, while the potential of terrestrial carbonate deposits as local climate archives had yet to be realized.
The road to the climate archive: Soreq Cave
In an interview with the Davidson Institute website, Bar-Matthews said that the idea that cave deposits might serve as a climate archive was first proposed in the 1980s. Researchers studying such deposits in northwestern England and the Canadian Rockies realized that, because they form only when water drips into a cave, they reflect wet climatic periods rather than frozen ones.
At the time, however, technical difficulties in dating the deposits and interpreting their isotopic composition limited researchers’ ability to extract reliable climate information from them. That same group even published a paper arguing that this kind of research was too expensive, especially when it came to precise dating, and that the investment was not justified.
5 View gallery


During the study, Bar-Matthews examined cross-sections of stalactites that revealed growth rings similar to those found in trees. Cross-section of a stalactite
(Photo: Shutterstock, Lapis2380)
Against this backdrop, in the early 1990s, Bar-Matthews, together with Dr. Avner Ayalon of the Geological Survey of Israel, began studying cave deposits in Soreq Cave near Jerusalem. According to Bar-Matthews, the cave was about to open to the public, and the Nature Reserves Authority approached the Geological Survey to conduct research that could help preserve the cave’s beauty and uniqueness in light of the expected rise in visitor numbers. During the study, Bar-Matthews examined cross-sections of stalactites that revealed growth rings—similar to tree rings—of varying thickness and color.
“It was clear to me that each growth layer might contain stored information,” she said. “I understood that the drop of water from which such a variety of rings precipitated must preserve a composition bearing an environmental ‘signature.’” This insight marked the beginning of a long process that ultimately led to the creation of a climate archive from cave stalactites.
To understand that environmental signature, Bar-Matthews and her colleagues began investigating the relationship between the isotopic composition of carbon and oxygen and the amount of rainfall. Their work produced two key papers, published in 1991 and 1996, which showed that the isotopic composition of stalactites directly reflects the composition of rainwater and soil water at the time of their formation. These studies established the link between the isotopic composition of cave deposits and the climatic conditions in the surrounding environment.
The instrument that paved the way
The findings showed that stalactites and stalagmites preserve a reliable record of past climate conditions, but the ages of the recorded periods had not yet been established. Bar-Matthews therefore turned to the development of precise dating methods together with colleagues in Israel and the United States, especially Aaron Kaufman of the Department of Environmental Sciences at the Weizmann Institute of Science.
Dating stalactites is based on the decay of uranium: when calcium carbonate precipitates from a drop of water and forms a new layer on a stalactite, it contains trace amounts of uranium but almost no thorium. Over time, the uranium decays at a known rate into thorium. Measuring the uranium-thorium ratio with a dedicated mass spectrometer makes it possible to calculate the age of each layer, sometimes with an accuracy of a few hundred years, even tens of thousands of years into the past. This makes it possible to construct a continuous timeline of a stalactite’s growth.
Initial dating using the method developed by Bar-Matthews and her colleagues was carried out with mass spectrometers at international research institutes and yielded high-resolution ages for cave formations. In the early 2000s, the Geological Survey of Israel acquired a dedicated mass spectrometer and became a national leader in uranium-thorium dating.
Using this precise dating method, studies showed that the deposits in Soreq Cave formed continuously through glacial and interglacial periods, and that the isotopic changes they recorded reflect climatic shifts over time. Similar patterns were later identified in other caves across Israel, confirming that the findings from Soreq represent a regional, not merely local, archive.
5 View gallery


Thanks to the work of Bar-Matthews and her colleagues, Soreq Cave became one of the world’s most important paleoclimate sites. The stalactites of Soreq Cave
(Photo: Wikimedia, Relly.hendel)
Comparisons with deposits from the Mediterranean Sea revealed simultaneous isotopic changes in both cave and marine records, leading to the conclusion that the isotopic signature preserved in stalactites reflects a global pathway, beginning in seas and oceans, passing through clouds, rain and soil, and ultimately reaching the cave. Precise dating thus turned cave deposits into an especially reliable tool for reconstructing the timing, pace and magnitude of climate change on land, and for understanding its effects on the environment and on human societies.
Thanks to the work of Bar-Matthews and her colleagues, especially Avner Ayalon, Soreq Cave became one of the world’s most important paleoclimate sites and a global reference archive against which data from other regions can be compared.
Later, as research into active tectonics advanced and interest grew in assessing seismic risk, Bar-Matthews expanded her work into the field of paleoseismology. The question arose whether stalactites—relatively delicate and fragile structures—might not only preserve climate archives, but also record evidence of significant earthquakes. Through multidisciplinary collaboration with earthquake researchers, ancient seismic events were identified through fractures, tilting and abrupt changes in stalactite growth. In this way, Bar-Matthews showed that ancient earthquakes can be detected and the seismic catalog extended thousands of years into the past.
The personal challenge and a message to future generations
Bar-Matthews said she began writing her doctoral dissertation after the birth of her third daughter and completed it shortly before the birth of her fourth child. “A colleague convinced me at the time that it was possible to do a PhD even as a mother of small children. Throughout my career, I had to juggle constantly; during working hours, I was extremely efficient—every minute and every hour mattered to me. In the afternoons, evenings and on weekends, we were a family—and we never forgot that.”
Scientifically, she sometimes encountered skepticism from colleagues, but in her words, “It was precisely the arguments, and perhaps even the insults I endured, that strengthened my belief in what I was doing. I felt like a missionary trying to convince scientists that this research was groundbreaking.”
Today, her work is considered a milestone in paleoclimate research. Her research showed that cave deposits can serve as a key tool for understanding climate change and for reconstructing the climate history of terrestrial regions with unprecedented precision. In a message to the next generation, she concludes: “Earth sciences are an endless puzzle, and those who have the privilege of being part of it keep assembling it every day.”

