One of the central issues in negotiations between the United States and Iran is the fate of enriched uranium, a material critical to both nuclear energy and weapons.
Over the weekend, U.S. President Donald Trump addressed reports that Iran had buried enriched uranium underground. “We’re going to get it together. We’ll go in with Iran, at a slow pace, go down and start digging with heavy equipment. We’ll bring it back to the United States,” he said. Iran, for its part, denied that any agreement exists regarding transferring uranium.
The dispute highlights a broader question: what uranium enrichment is, why it is difficult to achieve and how it relates to the ability to harness the vast energy stored in atomic nuclei.
Uranium is a heavy, radioactive metallic element, with atomic number 92, meaning its nucleus contains 92 protons. It is the heaviest element found naturally in significant quantities and is relatively abundant, with reserves expected to last for many years, especially if extraction from seawater becomes viable.
Under certain conditions, uranium can undergo nuclear fission, a process in which the nucleus splits into smaller nuclei, releasing a large amount of energy. This differs from natural radioactive decay, in which particles or electromagnetic radiation are emitted spontaneously.
The nucleus of an atom contains protons and neutrons. While the number of protons defines the element, the number of neutrons can vary, creating different isotopes. Uranium has several isotopes, but the most important for nuclear applications is uranium-235, which contains 92 protons and 143 neutrons.
When a neutron strikes a uranium-235 nucleus at the right speed, there is a high probability the nucleus will split, releasing energy and additional neutrons. These neutrons can strike other uranium-235 nuclei, creating a self-sustaining chain reaction. Materials capable of sustaining such reactions are known as fissile.
However, uranium-235 makes up less than 1% of naturally occurring uranium. More than 99% consists of uranium-238, which has 92 protons and 146 neutrons. While uranium-238 can undergo fission under certain conditions, it cannot sustain a chain reaction, making it unsuitable for nuclear weapons.
With sufficient fissile material, a chain reaction can escalate rapidly, producing an enormous number of fissions in less than a millionth of a second, as in a nuclear explosion. Alternatively, the reaction can be controlled, as in nuclear reactors, which use the heat generated to produce electricity.
The rate and intensity of a chain reaction depend on the amount and concentration of fissile material. A higher concentration increases the likelihood that neutrons will trigger additional fissions.
This leads to the need for uranium enrichment — the process of increasing the proportion of uranium-235 relative to uranium-238. In natural uranium, uranium-235 accounts for only about 0.7%, too low for most practical applications.
For nuclear power plants, enrichment to about 3% to 5% uranium-235 is typically sufficient, though some reactors require up to 20%. In contrast, nuclear weapons require much higher levels. Uranium enriched to 20% or more is considered weapons-usable, though practical weapons typically use uranium enriched above 90%.
The atomic bomb dropped on Hiroshima in 1945 used uranium enriched to about 80%. Higher enrichment levels would have produced a more powerful explosion.
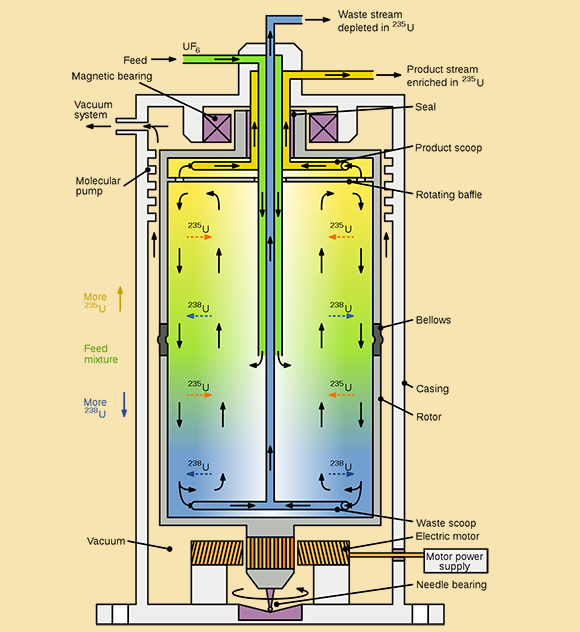
Separating uranium isotopes is technically challenging because their chemical properties are nearly identical. The process relies on slight differences in physical properties, particularly mass, with uranium-235 being only about 1.5% lighter than uranium-238.
Most enrichment methods require uranium to be converted into a gas, uranium hexafluoride, which is toxic and corrosive but easier to process. The most common method today uses centrifuges — rapidly spinning cylinders that separate isotopes based on mass. The heavier uranium-238 moves outward, while lighter uranium-235 concentrates toward the center.
Another method, gas diffusion, allows uranium hexafluoride gas to pass through membranes with tiny holes. Lighter molecules move slightly faster, resulting in gradual enrichment. However, the differences are extremely small, requiring thousands of stages to achieve significant enrichment.
Because each step yields only a slight increase in uranium-235 concentration, enrichment requires large cascades of centrifuges or repeated diffusion stages. The process consumes significant amounts of energy and is costly.
As a byproduct, enrichment produces depleted uranium, which consists mostly of uranium-238. Though not useful for nuclear reactors or weapons, it has applications in civilian and military contexts, including radiation shielding and armor-piercing munitions, due to its high density.
Globally, enriched uranium is essential both for nuclear energy and military purposes. Most of the roughly 500 nuclear power plants worldwide use low-enriched uranium. The ability to enrich uranium is therefore closely monitored, as the transition from civilian to military use can be relatively rapid.
Countries possessing nuclear weapons typically also have enrichment capabilities. Others, including Germany, the Netherlands and Japan, can enrich uranium but do not use it for weapons. Russia is a leading producer, responsible for about half of global enrichment capacity, followed by multinational and state-owned firms in Europe, China and France.
The five major nuclear powers — the United States, Russia, China, France and Britain — produce little new highly enriched uranium for weapons, relying instead on stockpiles built during the Cold War. In many cases, highly enriched uranium from dismantled weapons has been diluted for use as reactor fuel, a process seen as a major success in nuclear disarmament.
Iran’s nuclear program includes large-scale enrichment facilities. According to open-source reports, uranium is converted into uranium hexafluoride gas at a facility in Isfahan before being sent to enrichment sites in Natanz and Fordo. Natanz is the largest facility, while Fordo is built deep underground.
Reports indicate Iran has produced hundreds of kilograms of uranium enriched to about 60%, a level from which further enrichment to weapons-grade can be achieved relatively quickly. Some estimates suggest Iran could produce enough material for a nuclear weapon within weeks, with total stockpiles potentially sufficient for multiple weapons.
Iran has spent more than four decades developing its enrichment technology. According to reports, Israeli strikes during Operation Rising Lion damaged facilities in Natanz and Isfahan, though Fordo remained intact due to its fortified underground location.
Beyond uranium, isotope separation is used on a smaller scale for other elements, including hydrogen and lithium, with applications ranging from scientific research to nuclear technology. In lighter elements, larger relative mass differences make separation easier using chemical methods.
While isotope separation has played a major role in nuclear weapons development, it also has civilian applications, including in medicine, research and potentially future energy production through nuclear fusion.
The broader lesson is that enrichment technology sits at the intersection of civilian energy and military capability — a dual-use process that remains central to global security and diplomacy.