(Weizmann Institute of Science)
A team of Israeli researchers from the Weizmann Institute of Science, succeeded for the first time, to create a synthetic embryo model from stem cells cultured in a lab without eggs, sperm, or a human womb. According to a study. The method opens new horizons for studying stem cells from various organs in the developing embryo, and may one day make it possible to grow tissues and organs for transplantation using synthetic embryo models.
digital baby
(Powered by ACT News )
More stories:
Researchers created a cluster of cells that closely resembles human embryonic cells using only stem cells from lab mice. Earlier efforts did not succeed in creating credible models.
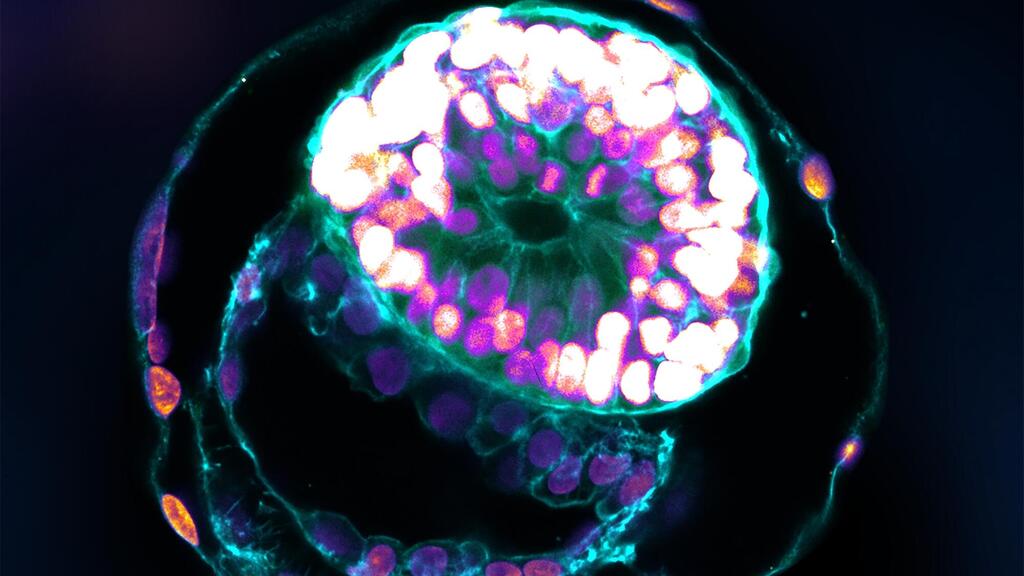
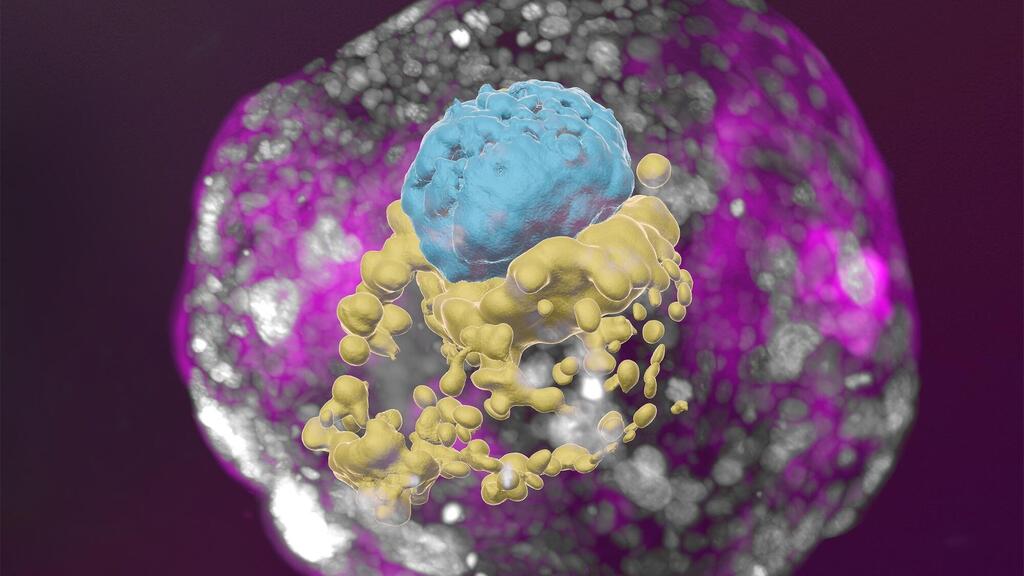
A research team headed by Prof. Jacob Hanna at the Weizmann Institute of Science has created complete models of human embryos from stem cells cultured in the lab – and managed to grow them outside the womb up to day 14. These synthetic embryo models had all the structures and compartments characteristic of this stage, including the placenta, yolk sac, chorionic sac and other external tissues that ensure the models’ dynamic and adequate growth.
According to a statement released by the Weizmann Institute, little is known about the early embryo because it is so difficult to study, for both ethical and technical reasons, yet its initial stages are crucial to its future development. During these stages, the clump of cells that implants itself in the womb on the seventh day of its existence becomes, within three to four weeks, a well-structured embryo that already contains all the body organs.
“The drama is in the first month, the remaining eight months of pregnancy are mainly lots of growth,” Hanna says. “But that first month is still largely a black box. Our stem cell–derived human embryo model offers an ethical and accessible way of peering into this box. It closely mimics the development of a real human embryo, particularly the emergence of its exquisitely fine architecture,” Professor Hana said.
Their study implied that their models faithfully emulated the process by which an early embryo gains all the structures it needs for beginning its transformation into a fetus. “Many failures of pregnancy occur in the first few weeks, often before the woman even knows she’s pregnant,” Hanna said. “That’s also when many birth defects originate, even though they tend to be discovered much later. Our models can be used to reveal the biochemical and mechanical signals that ensure proper development at this early stage, and the ways in which that development can go wrong.”
In fact, the study has already produced a finding that may open a new direction of research into early pregnancy failure. The researchers discovered that if the embryo is not enveloped by placenta-forming cells in the right manner at day 3 of the protocol (corresponding to day 10 in natural embryonic development), its internal structures, such as the yolk sac, fail to properly develop.
“An embryo is not static. It must have the right cells in the right organization, and it must be able to progress – it’s about being and becoming,” Hanna said. “Our complete embryo models will help researchers address the most basic questions about what determines its proper growth.
Hanna said there was a moral component to his work. "This approach is extremely valuable because it could, to a large extent, bypass the technical and ethical issues involved in the use of natural embryos in research and biotechnology," he said.
According to the scientists, the discovery may help researchers understand why early miscarriages occur and what causes congenital defects that may appear at the beginning of pregnancy. It could also help in understanding how to create organs for transplantation.
The model created by the Weizmann Institute team could have significant implications in the fields of science and medicine and could provide unprecedented insight into the early stages of embryonic development.
SYNTHETIC EMBRYO MODELS OPEN NEW AVENUES IN MEDICAL SCIENCE